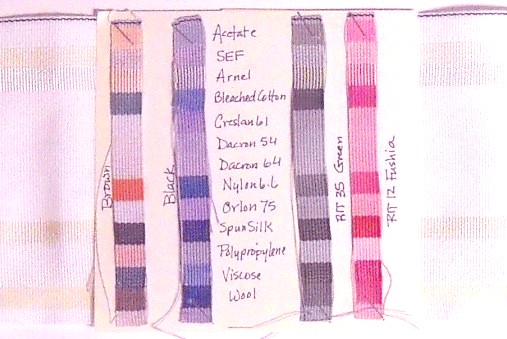
When chemists want to study how dyes interact with various fibers they use a “multi-fiber” ribbon. Each band of this ribbon is composed of a different fiber. We soaked strips of white multi-fiber ribbon in four different dyes, a brown dye and a black dye which were mixtures of several individual pigments, and a pink and a green dye which had one pigment component each. The various fibers interacted with assorted pigments in the dye mixtures, producing strips with multicolor bands. The single component dyes produced monochromatic strips with bands of different intensity.
We considered the origin and looked at the chemical structure of each fiber. Cotton is cellulose from plants; acetate, Arnel and viscose (rayon) are chemically modified forms of cellulose. Wool and silk are proteins from animals. The rest are synthetic polymers ultimately derived from petrochemicals including polypropylene (PP, resin identification code 5), Dacron (PET, polyethylene terephthalate, resin identification code 1), SEF (self extinguishing fabric), Creslan and Orlon (all acrylonitrile), and nylon. Would you expect fibers from similar sources to behave similarly with respect to the dyes? Did they?
Students dyed samples each of ten different unknown fabric swatches in each of the four dye solutions. (Wednesday students prepared samples for Monday and Tuesday). They compared the colors produced with the bands on the dyed multi-fiber ribbon to determine which fiber composed each unknown fabric.
We considered the origin and looked at the chemical structure of each fiber. Cotton is cellulose from plants; acetate, Arnel and viscose (rayon) are chemically modified forms of cellulose. Wool and silk are proteins from animals. The rest are synthetic polymers ultimately derived from petrochemicals including polypropylene (PP, resin identification code 5), Dacron (PET, polyethylene terephthalate, resin identification code 1), SEF (self extinguishing fabric), Creslan and Orlon (all acrylonitrile), and nylon. Would you expect fibers from similar sources to behave similarly with respect to the dyes? Did they?
Students dyed samples each of ten different unknown fabric swatches in each of the four dye solutions. (Wednesday students prepared samples for Monday and Tuesday). They compared the colors produced with the bands on the dyed multi-fiber ribbon to determine which fiber composed each unknown fabric.
We also investigated various commercial acid-base indicators, and tested homemade indicator paper made by soaking heavy paper in hot juices from fruits, vegetables and spices. We discovered that the pink/purple papers (beet, cranberry, blueberry, red cabbage and grape) turned greenish or bluish in the presence of base. These colors are due to anthocyanins which are pink in acidic solutions (pH < 7), purplish in neutral solutions (pH ~ 7), and greenish-yellow in alkaline (basic) solutions (pH > 7). Curcumin, the compound that gives turmeric its bright yellow color, turned dark orange in base.
You and your family can try making your own indicators, and explore other foods and plants in your kitchen and garden. For some suggestions, check the site “Home and Garden pH Indicators, Common Household Items” at http://chemistry.about.com/cs/acidsandbases/a/aa060703a.htm.